| BHM’s drives success through Pharmacy and Medical Necessity Reviews and provides this report summary of Prescription Drug Monitoring Programs success stories as efficient and financially impactful examples. BHM’s review services will add to your organization’s efforts. Click HERE to schedule a call with one of our Review Services Experts. |
 President Trump said, “The opioid crisis is an emergency, and I’m saying officially right now it is an emergency. It’s a national emergency. We’re going to spend a lot of time, a lot of effort and a lot of money on the opioid crisis.” One type of program payers and providers can both support are prescription drug monitoring programs (PDMPs). PDMPs combine the downstream benefits of sharing care data which improves care, monitors drug use, and discourages fraud.
President Trump said, “The opioid crisis is an emergency, and I’m saying officially right now it is an emergency. It’s a national emergency. We’re going to spend a lot of time, a lot of effort and a lot of money on the opioid crisis.” One type of program payers and providers can both support are prescription drug monitoring programs (PDMPs). PDMPs combine the downstream benefits of sharing care data which improves care, monitors drug use, and discourages fraud.
The CDC released a report Integrating & Expanding Prescription Drug Monitoring Program Data: Lessons from Nine States detailing a promising strategy for addressing the prescription opioid overdose epidemic. The study focused on improving the use of PDMPs.
PDMPs are state-run databases that collect patient-specific prescription information at the point of dispensing. Data are transmitted to a central repository where, in most states, authorized users such as medical professionals, public health agencies, regulatory bodies, and law enforcement agencies may access them.
Many states have promoted use of PDMPs by registered prescribers and dispensers to inform their clinical decisions and allow for intervention at the point of care. However, PDMP data that have not been well integrated into health information technology (HIT) systems at the point of care for efficient workflow, coupled with limited data sharing across states, have slowed adoption of PDMP use among health care professionals who prescribe and dispense prescription opioids.
To increase use of PDMPs and to effectively reduce prescription opioid misuse and overdose, the Substance Abuse and Mental Health Services Administration (SAMHSA) funded projects in nine states from fiscal years 2012 to 2016 through its PDMP Electronic Health Records (EHRs) Integration and Interoperability Expansion (PEHRIIE) program.
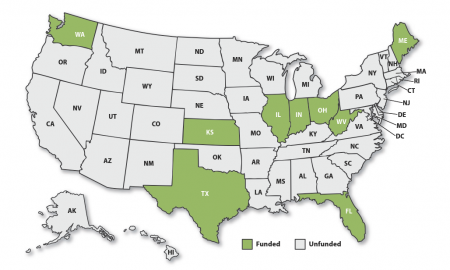
The project states were: Florida, Illinois, Indiana, Kansas, Maine, Ohio, Texas, Washington State, and West Virginia.
Prescription Drug Monitoring Programs Lessons
Snapshot of the lessons:
- Learn from the field.
- Start small, then expand.
- Consider state context.
- Engage stakeholders early and often.
- Design a mechanism for monitoring progress.
Each of the key lessons for program design and implementation is further described below:
1. Learn from the field. Working with multi-state vendors enhanced progress; efficiencies in multi-state initiatives such as integration are realized when multiple states work with the same technical partners, including PDMP vendors, EHR vendors, and pharmacies. For example, a rapidly increasing knowledge base in interstate data sharing was being built by users of the PMPi hub as use of that hub increased from its start in 2011. That knowledge base enabled five of the six PEHRIIE cohort one states engaging in two-way interstate data sharing to greatly expand interstate sharing during the project, whether they had already initiated interstate sharing prior to the project (Indiana and Ohio) or initiated interstate sharing as part of the project (Illinois, Kansas, and West Virginia). In particular, use of the PMPi hub facilitated interstate sharing for these states with most of their bordering states—an important data source for providers seeking a comprehensive prescription history for their patients.
In addition, Kansas, Ohio, and West Virginia achieved integration with the Kroger pharmacy chain (though Kroger’s work in Kansas was not a part of the PEHRIIE project). In operating in multiple states, Kroger strongly favored an integration solution that could apply to multiple states, rather than working out separate solutions with individual states.
2. Start small, then expand. Building upon early foundation pilots and leveraging existing HITs is important. Five states (Illinois, Indiana, Kansas, Ohio, and Washington) built on work from an earlier pilot funded by ONC and SAMHSA, Enhancing Access to PDMPs using Health Information Technology, which provided a substantial foundation for the PEHRIIE activities. The ONC-SAMHSA pilot project was designed to improve prescriber access to PDMP data through secure report messaging, enhancing EHR systems, and exploring opportunities to leverage hubs for transferring PDMP data. Experience with the earlier pilot substantially enhanced each of these states’ ability to carry out a more fully-developed or expanded interoperability project as part of PEHRIIE.
3. Consider state context. A wide range of state features and ongoing activities affected the progress of the work. States chose approaches largely due to contextual factors such as existing state infrastructure and regulatory mandates. Figure 5 lists some factors that should be considered as the program is designed and implemented.
4. Engage stakeholders early and often. The Illinois PDMP was able to engage physicians at Anderson Hospital in recognizing the importance of streamlined access to PDMP reports through integration with Anderson’s EHR system. These physicians were critical in persuading the hospital administration to continue the project from its initial pilot integration with the ONC-SAMHSA pilot program. In Texas, the state brought stakeholders together to form the Technical Work Group, a collaborative work group that has pulled relevant parties together and provided the financial stability needed to drive project implementation.
Similar to Texas, prior to the launch of the PDMP program Washington initiated relationship building, which has been credited as a key to the success of the state’s PEHRIIE work. Staff of Washington’s PDMP, Prescription Review, were able to engage numerous stakeholders, including the Washington chapter of the American College of Emergency Physicians (ACEP), the Washington State Hospital and Medical Associations, the Washington Unintentional Poisoning Workgroup, and the Washington “ER is for Emergencies” Work Group. Prescription Review staff also worked closely with their PDMP vendor, HID, and the vendors for the OHP HIE and EDIE hub, laying the foundation for PDMP-EHR integration in the state. Engaging key stakeholders in this way has yielded strong, sustainable results in both states, and stakeholders have continued to actively work together to integrate the states’ PDMP into EHRs.
5. Design a mechanism for monitoring progress. For each of the nine states, the PEHRIIE project was part of a larger vision and set of objectives the state intended to accomplish. To monitor the progress of each objective, including those associated with PEHRIIE, it is important to have a plan for measuring progress that helps ensure the agency level buy-in needed to sustain the project and that provides the basis for future cross-agency efforts. While state stakeholders were clearly familiar with standard PDMP metrics, having a plan up front with benchmarks to monitor use of PDMP data once integration and interoperability is established and adopted is crucial. PDMPs varied in their ability to determine providers associated with specific integration and interoperability sites, such as an EHR system or an HIE.
A second reason to establish a mechanism for documenting and monitoring progress is to support project continuity in the event of changes in key personnel. In several states, due to turnover and changing priorities of stakeholders and state agency personnel, the program was stalled for periods of time. Having a documented plan and proposed timeline endorsed by all parties will promote seamless transitions as individuals leave and vendors or priorities change.
BHM’s drives success through Pharmacy and Medical Necessity Reviews and provides this report summary of Prescription Drug Monitoring Programs success stories as efficient and financially impactful examples. BHM’s review services will add to your organization’s efforts. Click HERE to schedule a call with one of our Review Services Experts.